 
Thus, looking at the activation parameters can reveal a lot about what is going on in the transition state. Dissociative pathway: one molecule converts to two molecules in rate determining step, greater degrees of freedom in two independently moving molecules, entropy increases.Dissociative pathway: more bond breaking in rate determining step, higher enthalpy needs.these two molecules have more degrees of freedom in which to partition their energy than they did as one molecule. However, the molecule breaks into two molecules in the rate determining step. 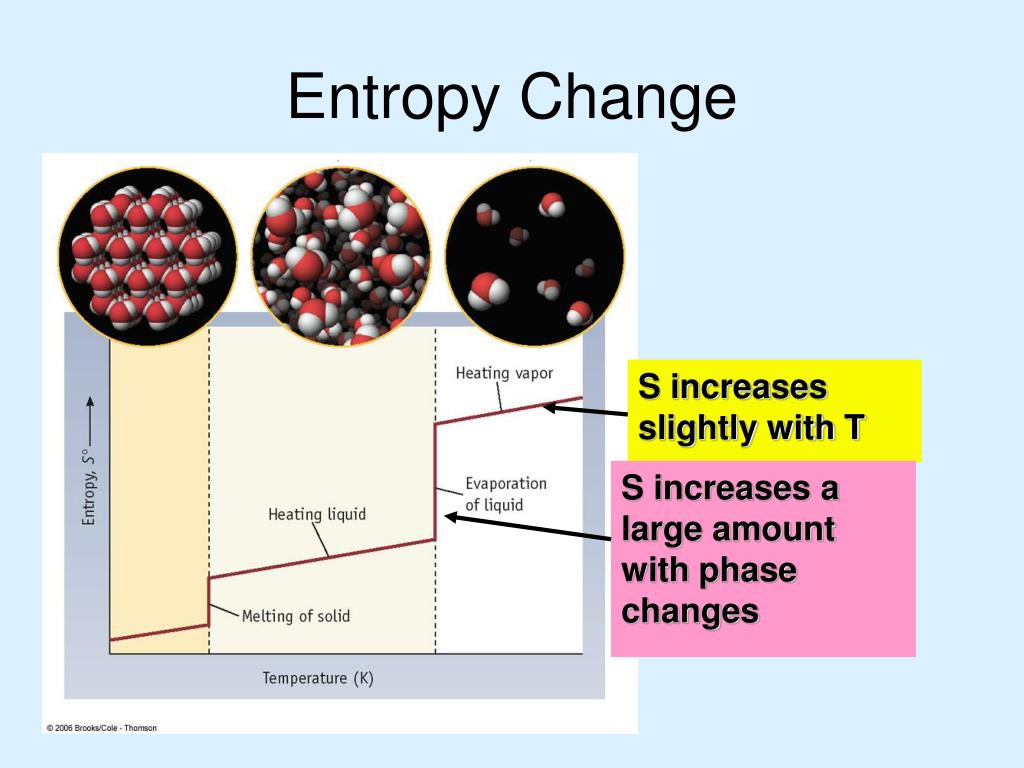
The higher enthalpy of activation results because a bond must be broken in the rate determining step. On the other hand, the dissociative pathway is marked by a higher enthalpy of activation but a positive entropy of activation. A positive entropy change indicates that the reaction is becoming more disordered, which is more thermodynamically stable than a reaction where entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
